“Acetone Extra Pure” has been added to your cart. View cart
Back to products


Ferric Chloride Anhydrous Extra Pure
$ 18.00 Original price was: $ 18.00.$ 17.90Current price is: $ 17.90.
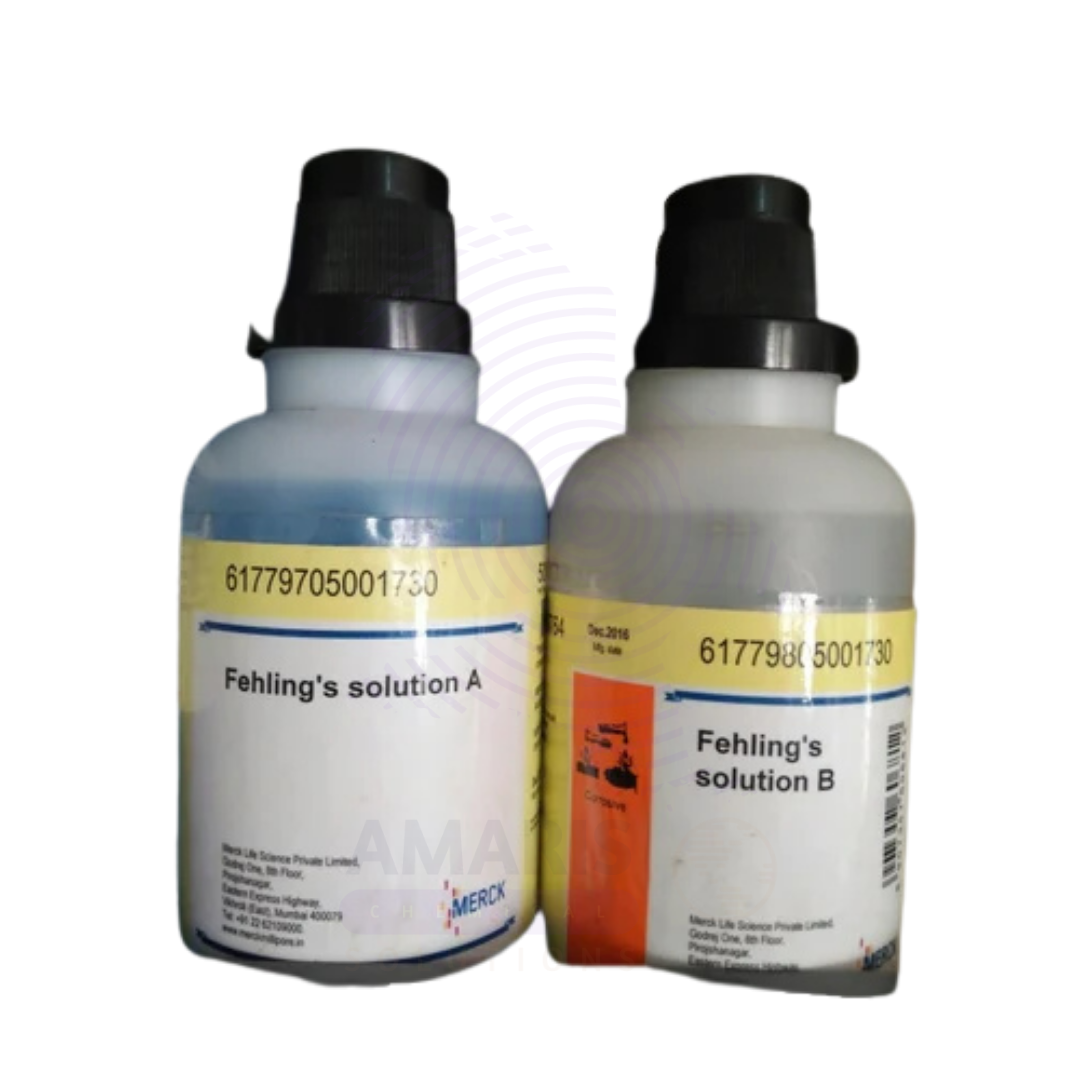
Fehling Solution 1 and 2 Extra Pure
$ 36.00 Original price was: $ 36.00.$ 35.90Current price is: $ 35.90.
Whatsapp Order
Fehling Solution 1 and 2 Extra Pure are essential reagents in classical laboratory chemistry, particularly used for the qualitative detection of reducing sugars such as glucose.
- Fehling Solution 1 contains copper(II) sulfate, while
- Fehling Solution 2 contains an alkaline potassium sodium tartrate (Rochelle salt) solution.
When mixed in equal parts just before use, they form a deep blue complex that, upon heating with a reducing sugar, yields a brick-red precipitate of cuprous oxide, confirming the presence of aldehyde groups.
These solutions are highly valuable in educational labs, food testing, and biochemical experiments. Store them separately in cool, dry conditions, and always mix fresh for accurate results.
Description
Table of Contents
Toggle
Fehling Solution 1 and 2 Extra Pure
Primary Uses
- Detection of reducing sugars:
Widely used in qualitative analysis to test for the presence of aldehyde functional groups, especially in reducing sugars like glucose. - Differentiation between aldehydes and ketones:
Reacts with aldehydes to form a red precipitate of copper(I) oxide, while ketones generally do not react. - Educational experiments in redox chemistry:
Demonstrates redox reactions and the principle of oxidation-reduction using visible color change.
Secondary Uses
- Identification of carbohydrate content in unknown samples:
Used to confirm the presence of reducing sugars in plant extracts, juices, or hydrolyzed polysaccharides. - Routine qualitative analysis in food chemistry labs:
Applied in basic food testing kits and protocols to check sugar levels in various food products. - Historical method reference in pharmaceutical analysis:
Though mostly replaced by modern techniques, still referenced in classic pharmaceutical and biochemical protocols.
KEY PRODUCT FEATURES
KEY ATTRIBUTES
1. Basic Identification Attributes
- Chemical Name: Fehling Solution (Part I & II)
- Components:
- Fehling’s Solution I: Aqueous copper(II) sulfate (CuSO₄·5H₂O) solution
- Fehling’s Solution II: Alkaline sodium potassium tartrate (Rochelle salt) in sodium hydroxide solution
- Grade: Extra Pure (Laboratory Reagent Grade)
- Appearance:
- Solution I: Clear blue solution
- Solution II: Colorless or pale solution
2. Physical & Chemical Properties
- Fehling’s Solution I:
- Copper(II) sulfate concentration: Typically ~0.5 M
- Fehling’s Solution II:
- Sodium potassium tartrate: Chelates the copper ions
- Sodium hydroxide concentration: ~1.5 M
- When mixed (equal volumes of I & II): Deep blue complex due to formation of a copper-tartrate complex
3. Safety & Hazard Attributes
- GHS Classification:
- Skin irritation (Category 2)
- Eye irritation (Category 2A)
- Harmful to aquatic life
- Hazard Statements:
- H315: Causes skin irritation
- H319: Causes serious eye irritation
- H411: Toxic to aquatic life with long lasting effects
- PPE Requirements:
- Gloves (nitrile recommended)
- Protective goggles
- Lab coat
- Use in well-ventilated area or fume hood
- First Aid Measures:
- Inhalation: Move to fresh air
- Skin Contact: Wash with soap and water
- Eye Contact: Flush with water for several minutes
- Ingestion: Rinse mouth, seek medical attention
4. Storage & Handling Attributes
- Storage Conditions:
- Store in tightly closed containers
- Protect from light and heat
- Store components separately until ready for use
- Handling Notes:
- Avoid contact with eyes, skin, and clothing
- Mix just before use to ensure activity
5. Regulatory & Compliance Attributes
- Component CAS Numbers:
- Copper(II) sulfate pentahydrate: 7758-99-8
- Sodium potassium tartrate: 6381-59-5
- Sodium hydroxide: 1310-73-2
- EC Numbers:
- CuSO₄·5H₂O: 231-847-6
- Hazard Class (mixed): 8 (Corrosive)
- UN Numbers (if shipped mixed): UN 3264
6. Laboratory Applications
- Primary Uses:
- Qualitative test for reducing sugars (e.g., glucose, fructose, maltose)
- Used in school and biochemical labs for carbohydrate analysis
- Secondary Uses:
- Demonstrations in redox chemistry
- Educational reagent in classical qualitative analysis
SAFETY HANDLING PRECAUTIONS
SAFETY MEASURES
SAFETY & HANDLING
- Corrosive – due to NaOH in Solution 2
- Toxic to aquatic life – due to copper sulfate
- Use gloves, goggles, and lab coat
- Storage:
- Store separately in tightly closed bottles
- Keep in a cool, dry place away from light and incompatible substances
FIRST AID MEASURES
- Inhalation: Move to fresh air, seek medical attention if irritation persists
- Skin/Eye Contact: Rinse immediately with water for at least 15 minutes
- Ingestion: Rinse mouth, do not induce vomiting—seek urgent medical care
Related products
Alizarin Extra Pure
Alizarin Extra Pure is a high-grade anthraquinone-based dye, presented as a reddish-brown crystalline powder, renowned for its vivid coloration and strong affinity for metal ions. It is primarily used in analytical chemistry as a complexometric indicator, especially for detecting aluminum, calcium, and other metal ions through color change. Due to its exceptional purity, it is also employed in histological staining, textile research, and pigment synthesis where precision and consistency are critical. Alizarin Extra Pure offers excellent stability and intense coloration, making it ideal for scientific, educational, and specialized industrial applications. Proper storage in a cool, dry environment is essential to maintain its effectiveness and prevent degradation.
Aluminium Ammonium Sulphate Extra Pure
Aluminium Ammonium Sulphate Extra Pure, also known as ammonium alum, is a high-purity double salt composed of aluminum sulfate and ammonium sulfate, typically appearing as colorless or white crystalline granules. Valued for its excellent stability and solubility in water, it is widely used in analytical chemistry, dye mordanting, and laboratory reagent preparations. Its ability to precipitate proteins makes it useful in biochemical applications, while its astringent and antimicrobial properties support its role in pharmaceutical and cosmetic formulations. The extra pure grade ensures minimal impurities, making it suitable for high-precision scientific work. It should be stored in a tightly sealed container in a dry, cool area to prevent moisture absorption and maintain its effectiveness.
Aluminium Nitrate Extra Pure
Aluminium Nitrate Extra Pure is a high-purity, white crystalline compound that is highly soluble in water and widely used in laboratory chemistry for analytical, synthesis, and research purposes. It serves as a reliable source of aluminum ions in solution, making it valuable in qualitative analysis, coordination chemistry, and materials science studies. This compound is often utilized in the preparation of other aluminum salts, as a nitrating agent, and in controlled oxidation reactions. Its extra pure grade guarantees minimal interference from contaminants, ensuring accurate, reproducible results in sensitive experimental work. Aluminium Nitrate should be stored in tightly sealed containers, away from moisture and incompatible substances, due to its oxidizing nature and hygroscopicity.
Aluminium Potassium Sulphate Hydrated Extra Pure
Aluminium Potassium Sulphate Hydrated Extra Pure, commonly known as potash alum, is a high-purity, colorless to white crystalline compound containing water of crystallization. It is widely used in laboratory settings for analytical chemistry, especially in the preparation of standard solutions, precipitation reactions, and as a source of aluminum ions. Its astringent and antiseptic properties also make it useful in biochemical and pharmaceutical experiments. The hydrated form offers improved handling and solubility in water, which supports its use in buffer systems and crystal growth studies. With minimal impurities, the extra pure grade ensures consistent and reliable results in sensitive research applications. It should be stored in a dry, cool environment in well-sealed containers to preserve its quality and prevent moisture loss.
Ammonia Solution Extra Pure
Ammonia Solution Extra Pure is a high-purity, clear, colorless liquid composed of ammonia gas dissolved in water, emitting a strong, characteristic pungent odor. Widely used in laboratory chemistry, it serves as a crucial reagent in acid-base titrations, complexometric analysis, and the preparation of ammonium salts and metal-ammonia complexes. Its alkaline nature makes it valuable for pH adjustment, cleaning of laboratory glassware, and as a reducing agent in certain analytical procedures. The extra pure grade ensures low levels of impurities, supporting reliable results in sensitive experimental work. Due to its volatility and corrosiveness, it should be handled in well-ventilated areas and stored in tightly sealed, chemically resistant containers.
Ammonium Bromide Extra Pure
Ammonium Bromide Extra Pure is a high-purity, white crystalline powder highly soluble in water, widely used in laboratory settings for analytical, photographic, and synthetic applications. It serves as a reliable source of bromide ions in chemical reactions and is commonly used in preparing photographic emulsions, corrosion inhibitors, and certain pharmaceuticals. In analytical chemistry, it is employed in qualitative and quantitative analysis involving halide ion detection. The extra pure grade ensures minimal contamination, making it ideal for sensitive research and high-precision work. To preserve its stability and prevent moisture uptake, it should be stored in well-sealed containers in a cool, dry environment.
Ammonium Cupric Chloride Extra Pure
- SHORT DESCRIPTION


 Acidulants
Acidulants Antioxidants
Antioxidants Nutraceutical Ingredients (food)
Nutraceutical Ingredients (food)
 Collectors
Collectors Dust Suppressants
Dust Suppressants Explosives and Blasting Agents
Explosives and Blasting Agents Flocculants and Coagulants
Flocculants and Coagulants Frothers
Frothers Leaching Agents
Leaching Agents pH Modifiers
pH Modifiers Precious Metal Extraction Agents
Precious Metal Extraction Agents
 Antioxidants(plastic)
Antioxidants(plastic) Colorants (Pigments, Dyes)
Colorants (Pigments, Dyes) Fillers and Reinforcements
Fillers and Reinforcements Flame Retardants
Flame Retardants Monomers
Monomers Plasticizers
Plasticizers Polymerization Initiators
Polymerization Initiators Stabilizers (UV, Heat)
Stabilizers (UV, Heat)
 Antifoaming Agents
Antifoaming Agents Chelating Agents
Chelating Agents Coagulants and Flocculants
Coagulants and Flocculants Corrosion Inhibitors
Corrosion Inhibitors Disinfectants and Biocides
Disinfectants and Biocides Oxidizing Agents
Oxidizing Agents pH Adjusters
pH Adjusters Scale Inhibitors( water)
Scale Inhibitors( water)
 Antioxidants(cosmetic)
Antioxidants(cosmetic) Emollients
Emollients Fragrances and Essential Oils
Fragrances and Essential Oils Humectants
Humectants Preservatives
Preservatives Surfactants(cosmetic)
Surfactants(cosmetic) Thickeners
Thickeners UV Filters
UV Filters
 Fertilizers
Fertilizers Soil Conditioners
Soil Conditioners Plant Growth Regulators
Plant Growth Regulators Animal Feed Additives
Animal Feed Additives Biostimulants
Biostimulants Pesticides (Herbicides, Insecticides, Fungicides)
Pesticides (Herbicides, Insecticides, Fungicides)
 Active Pharmaceutical Ingredients (APIs)
Active Pharmaceutical Ingredients (APIs) Excipients
Excipients Solvents(pharmaceutical)
Solvents(pharmaceutical) Antibiotics
Antibiotics Antiseptics and Disinfectants
Antiseptics and Disinfectants Vaccine Adjuvants
Vaccine Adjuvants Nutraceutical Ingredients (pharmaceutical)
Nutraceutical Ingredients (pharmaceutical) Analgesics & Antipyretics
Analgesics & Antipyretics
 Analytical Reagents
Analytical Reagents Chromatography Chemicals
Chromatography Chemicals Spectroscopy Reagents
Spectroscopy Reagents Molecular Biology Reagents
Molecular Biology Reagents Biochemical Reagents
Biochemical Reagents Inorganic and Organic Standards
Inorganic and Organic Standards Laboratory Safety Chemicals
Laboratory Safety Chemicals Specialty Laboratory Chemicals(Special Laboratory Equipment)
Specialty Laboratory Chemicals(Special Laboratory Equipment)
 Demulsifiers
Demulsifiers Hydraulic Fracturing Fluids
Hydraulic Fracturing Fluids Scale Inhibitors(oil)
Scale Inhibitors(oil) Surfactants(oil)
Surfactants(oil) Drilling Fluids
Drilling Fluids
 Dyes and Pigments
Dyes and Pigments Bleaching Agents
Bleaching Agents Softening Agents
Softening Agents Finishing Agents
Finishing Agents Antistatic Agents
Antistatic Agents
 Admixtures
Admixtures Waterproofing Agents
Waterproofing Agents Sealants and Adhesives
Sealants and Adhesives Curing Compounds
Curing Compounds Concrete Repair Chemicals
Concrete Repair Chemicals Anti-Corrosion Coatings
Anti-Corrosion Coatings
 Surfactants(cleaning)
Surfactants(cleaning) Builders
Builders Enzymes
Enzymes Solvents (Cleaning)
Solvents (Cleaning) Fragrances
Fragrances
 Electronic Chemicals
Electronic Chemicals Catalysts
Catalysts Lubricants
Lubricants Photographic Chemicals
Photographic Chemicals Refrigerants
Refrigerants Automotive chemicals
Automotive chemicals Pyrotechnic Chemicals
Pyrotechnic Chemicals
 Biodegradable Surfactants
Biodegradable Surfactants Bio-based Solvents
Bio-based Solvents Renewable Polymers
Renewable Polymers Carbon Capture Chemicals
Carbon Capture Chemicals Wastewater Treatment Chemicals
Wastewater Treatment Chemicals
 Pigments
Pigments Solvents(paint)
Solvents(paint) Specialty Coatings
Specialty Coatings Binders/Resins
Binders/Resins Additives
Additives Driers
Driers Anti-Corrosion Agents
Anti-Corrosion Agents Functional Coatings
Functional Coatings Application-Specific Coatings
Application-Specific Coatings
 Fresh Herbs
Fresh Herbs Ground Spices
Ground Spices Whole Spices
Whole Spices Spice Blends
Spice Blends Dried Herbs
Dried Herbs
 Leavening Agents
Leavening Agents Dough Conditioners
Dough Conditioners Flour Treatments
Flour Treatments Fat Replacers
Fat Replacers Decoratives
Decoratives Preservatives(baking)
Preservatives(baking)
 Plasticizers & Softeners
Plasticizers & Softeners Reinforcing Agents
Reinforcing Agents Adhesion Promoters
Adhesion Promoters Vulcanizing Agents
Vulcanizing Agents Antidegradants
Antidegradants Blowing Agents
Blowing Agents Fillers & Extenders
Fillers & Extenders Accelerators & Retarders
Accelerators & Retarders