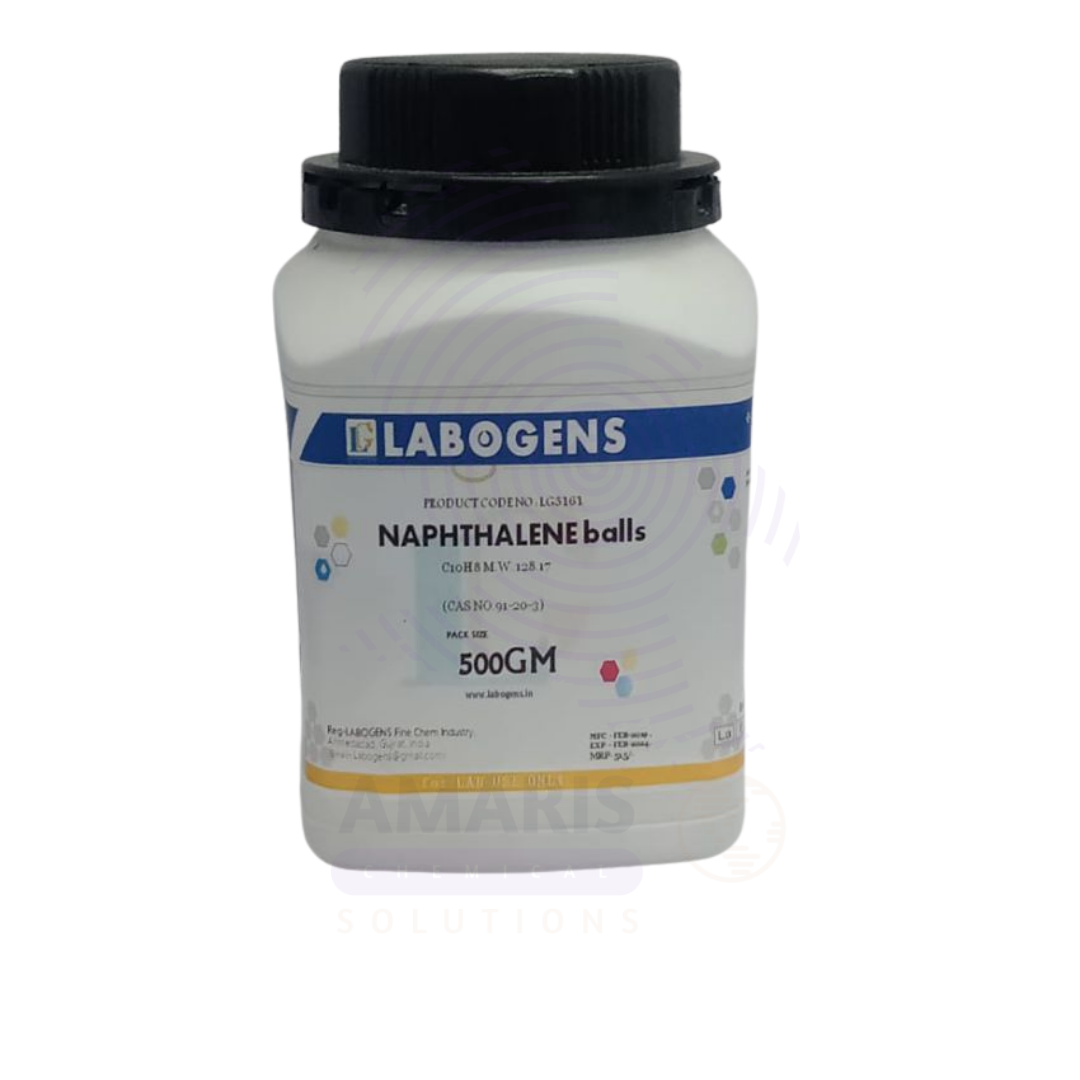
Naphthalene Balls Extra Pure
$ 20.09 Original price was: $ 20.09.$ 19.98Current price is: $ 19.98.
Naphthalene Balls Extra Pure are solid, white crystalline spheres composed of high-purity naphthalene (C₁₀H₈), refined for laboratory, industrial, and specialized household uses. Known for their strong, distinctive odor, these balls sublimate—changing directly from solid to vapor—making them exceptionally effective as fumigants and insect repellents, particularly against moths, silverfish, and mold in stored fabrics and books.
In laboratory settings, extra pure naphthalene balls are often used as a standard hydrocarbon compound for melting point determination, sublimation studies, and chemical synthesis. Their consistent purity and volatile nature also make them suitable for organic chemistry demonstrations and reactions. For safety, they should be handled in well-ventilated areas, kept away from flames or heat sources, and stored in airtight containers to minimize inhalation and fire risks.
Naphthalene Balls Extra Pure
Primary Uses
- Fumigant Insecticide for Mothproofing
- Used as a volatile repellent and toxicant in enclosed spaces (like wardrobes, storage chests, and garment bags) to protect woolens, furs, and other natural fibers from clothes moths (Tineola bisselliella) and carpet beetles. The subliming gas creates a lethal atmosphere for the larval stages of these pests.
- General Pest Deterrent in Domestic and Storage Settings
- Employed to repel a broader range of insects and other pests, including silverfish, cockroaches, and ants, from stored items such as books, documents, and bedding in cupboards, attics, and storage units.
- Deodorant and Air Freshener for Confined Spaces
- Used in bathrooms, lavatories, and musty basements to neutralize strong odors by masking them with its characteristic pungent aroma. Its continuous sublimation provides a long-lasting deodorizing effect in enclosed, unoccupied areas.
- Precursor in Organic Chemical Synthesis
- Serves as a fundamental raw material in the industrial synthesis of phthalic anhydride, which is a key intermediate for producing plastics (alkyl resins), dyes (azo dyes), and solvents. It is also used in labs to synthesize derivatives like sulfonated naphthalene.
Secondary Uses
- Teaching Tool in Demonstrating Sublimation
- An ideal substance for classroom or laboratory demonstrations of the physical process of sublimation (the direct transition from a solid to a gas), as it visibly vaporizes without melting at standard room temperature and pressure.
- Fuel Additive and Combustion Study
- Historically used as a volatile component in some solid fuel cubes (e.g., for military purposes or camping) due to its high flammability. It is used in educational settings to study combustion efficiency and soot formation due to its aromatic, carbon-rich structure.
- Solvent and Re-crystallization Standard
- In specific industrial processes, it acts as a solvent for some organic compounds. In teaching laboratories, its “extra pure” grade makes it an excellent substance for demonstrating the technique of purification via re-crystallization, given its well-defined crystal formation upon cooling.
- Preservative for Specimens and Taxidermy
- Used in museum collections and taxidermy as a deterrent against insect infestation of biological specimens, feathers, and mounted animals, helping to preserve their integrity in display cases and storage drawers.
| PACK SIZE | 500 grams Plastic Tin |
|---|
1. Basic Identification Attributes
- Chemical Name: Naphthalene
- CAS Number: 91-20-3
- HS Code: 2902.90.00
- Molecular Formula: C₁₀H₈
- Synonyms: Mothballs, Tar Camphor, White Tar, Naphthalin, Naphthene
2. Physical & Chemical Properties
- Physical State: Solid (white balls)
- Color & Odor: White; strong, characteristic aromatic odor
- Boiling Point: 218 °C
- Melting Point: 80.2 °C
- Density: ~1.14 g/cm³
- Solubility: Insoluble in water; soluble in organic solvents (ether, benzene, ethanol)
- pH Level: Not applicable (neutral organic compound)
- Vapor Pressure: 0.087 mmHg at 25 °C (sublimes easily)
- Flash Point: 79 °C (closed cup)
- Autoignition Temperature: 527 °C
- Viscosity: Not applicable (solid)
3. Safety & Hazard Attributes
- Hazard Class (GHS):
- Flammable solid (Category 2)
- Acute toxicity (Category 4)
- Carcinogenicity (Category 2)
- Hazardous to the aquatic environment – chronic (Category 1)
- NFPA Ratings:
- Health: 2
- Flammability: 2
- Reactivity: 0
- Exposure Limits:
- OSHA PEL: 10 ppm (50 mg/m³)
- ACGIH TLV: 10 ppm (skin)
- Reactivity: Stable under normal conditions; avoid heat, sparks, and oxidizers
4. Storage & Handling Attributes
- Storage Conditions: Store in a cool, dry, well-ventilated place away from heat or open flame
- Incompatible Materials: Strong oxidizing agents (e.g., nitric acid, chromates)
- Container Type: Tightly sealed containers made of glass or HDPE
- Shelf Life: Long shelf life if properly sealed and protected from heat
- Special Handling Requirements: Use gloves and safety goggles; avoid inhaling vapors and dust; ensure good ventilation
5. Regulatory & Compliance Attributes
- Regulatory Status:
- EPA: Listed as hazardous air pollutant (HAP)
- REACH: Registered
- OSHA: Regulated as hazardous
- DOT: Classified as hazardous material
- Hazard Symbols:
- Flame, Exclamation mark, Health hazard, Environment
- Transportation Restrictions:
- UN Number: UN1334
- Hazard Class: 4.1 (Flammable solid)
- Waste Disposal Method: Dispose of as hazardous waste in accordance with local regulations (RCRA guidelines)
6. Environmental & Health Impact
- Ecotoxicity: Highly toxic to aquatic organisms; long-term environmental damage possible
- Persistence in Environment: Moderately persistent; volatile but can accumulate in soil and water
- Carcinogenicity/Mutagenicity:
- IARC: Group 2B (possibly carcinogenic to humans)
- OSHA: Recognized as a potential occupational carcinogen
- Biodegradability: Low to moderate under aerobic conditions
SAFETY PRECAUTIONS
Personal Protective Equipment (PPE):
- Wear a lab coat, nitrile gloves, and chemical safety goggles.
- Use in a well-ventilated area or under a fume hood.
- Respiratory protection may be necessary in case of inadequate ventilation.
Handling:
- Avoid inhalation of vapors and dust.
- Prevent contact with skin and eyes.
- Wash hands thoroughly after handling.
Storage:
- Store in a cool, dry, and well-ventilated area away from heat and open flames.
- Keep the container tightly closed and away from food and drink.
- Store separately from oxidizing agents and acids.
FIRST AID MEASURES
Inhalation:
- Move the affected person to fresh air immediately.
- Seek medical attention if symptoms such as coughing, dizziness, or headache occur.
Skin Contact:
- Wash with soap and plenty of water.
- Remove contaminated clothing.
- Get medical attention if irritation or redness develops.
Eye Contact:
- Rinse cautiously with water for several minutes.
- Remove contact lenses if present and easy to do.
- Continue rinsing and seek medical attention if irritation persists.
Ingestion:
- Rinse mouth with water.
- Do NOT induce vomiting.
- Seek immediate medical advice—naphthalene can be toxic if swallowed.
FIRE FIGHTING MEASURES
Flammability:
- Flammable solid; may sublimate and form flammable vapors.
Extinguishing Media:
- Use dry chemicals, CO₂, or foam.
- Water spray may be used to cool surrounding containers.
Hazardous Combustion Products:
- May emit toxic fumes of carbon monoxide and carbon dioxide.
- Can release irritating or toxic fumes upon decomposition.
Firefighter Protection:
- Use self-contained breathing apparatus (SCBA) and full protective gear.
- Avoid inhalation of combustion fumes.
Related products
Cupric Oxide Extra Pure
Cupric Oxide Extra Pure is a black, fine powder extensively used in laboratory settings as a stable, high-purity source of copper in oxidation state +2. It is commonly applied in organic synthesis, inorganic reactions, and thermal decomposition studies, as well as in qualitative and quantitative analysis of copper. Its thermal stability also makes it suitable for ceramic glazing and as a precursor in catalyst preparation. This extra pure grade ensures low impurity levels, essential for accurate experimental results and reproducible data in both educational and research laboratories. It should be stored in a tightly sealed container away from moisture and strong acids.
Cupric Sulphate Pentahydrate Extra Pure
Cupric Sulphate Pentahydrate Extra Pure is a vibrant blue crystalline compound widely used in laboratories for its high solubility and consistent copper ion content. It is essential in analytical chemistry for Fehling's and Benedict’s solutions, in electrochemistry for plating and battery experiments, and as a biological nutrient in microbiological media. The pentahydrate form offers ease of handling and precise formulation in aqueous solutions. This extra pure grade ensures exceptional purity, making it suitable for teaching labs, research applications, and controlled experimental procedures. Store in a sealed container away from heat and moisture to maintain its crystalline integrity.
Magnesium Nitrate Hexahydrate Extra Pure
Magnesium Nitrate Hexahydrate Extra Pure is a crystalline compound known for its high solubility in water and exceptional purity, making it suitable for both laboratory and industrial applications. In analytical chemistry, it is commonly used as a reagent for preparing standard solutions and as a source of magnesium ions in various experimental procedures. Its oxidizing properties make it valuable in pyrotechnics and fertilizer formulations, where it provides essential nutrients for plant growth. This compound is also employed in ceramic production, textile treatments, and catalyst preparations, where a consistent, high-quality nitrate source is required. Its extra pure grade ensures minimal contamination, essential for high-precision work.
Magnesium Ribbon Extra Pure
Magnesium Ribbon Extra Pure is a highly reactive metallic strip known for its silvery appearance and clean-burning properties. It is widely used in laboratory demonstrations, particularly in combustion and oxidation experiments, where it burns with a bright white flame, showcasing exothermic reactions vividly. Due to its high purity, it ensures minimal contamination in analytical and research applications. Beyond academics, magnesium ribbon also finds niche uses in pyrotechnics, metal alloy production, and chemical synthesis where controlled reactivity and reliable performance are essential. Its lightweight and malleable nature make it easy to handle and ideal for precision experimental setups.
Manganese Metal Powder Extra Pure
Manganese Metal Powder Extra Pure is a fine, silvery-gray metallic powder known for its high reactivity and essential role in various chemical and metallurgical applications. In the laboratory, it is frequently used in redox reactions, alloy synthesis, and as a reagent for preparing manganese compounds. Its high surface area makes it particularly effective in catalysis and experimental procedures requiring fast reactivity. Industrially, manganese metal powder is integral to producing specialty steels and alloys, where it improves hardness, strength, and wear resistance. Its exceptional purity ensures consistent performance in research, metal refining, and advanced materials development.
Menthol Crystals Extra Pure
Menthol Crystals Extra Pure are colorless to white, needle-like crystals derived from peppermint or other mint oils and refined to the highest purity. Known for their strong, refreshing minty aroma, they are widely used in pharmaceutical, cosmetic, and personal care formulations. These crystals provide a cooling sensation on the skin and mucous membranes, making them a common ingredient in balms, liniments, throat lozenges, and inhalants. In laboratories, menthol crystals are also used in sensory studies and product development. Their purity ensures consistent performance and minimal interference in sensitive formulations.
Methanol Extra Pure
Methanol Extra Pure is a clear, colorless, and highly volatile liquid with a mild alcohol-like odor, refined to an exceptional level of purity. It is widely used as a solvent in laboratories and industrial settings due to its excellent miscibility with water and many organic compounds. Methanol serves as a key reagent in chemical synthesis, particularly in the production of formaldehyde, methyl esters, and biodiesel. In analytical chemistry, it’s used for chromatography and spectroscopic applications where high purity is essential. Its rapid evaporation rate and strong solvency make it ideal for cleaning delicate equipment and preparing lab samples.
Methyl Orange Disodium Salt Extra Pure
Methyl Orange Disodium Salt Extra Pure is a high-purity pH indicator commonly used in titrations and analytical chemistry to determine acidity levels. It exhibits a sharp color change from red in acidic environments to yellow in alkaline conditions, typically within the pH range of 3.1 to 4.4. This disodium salt form ensures excellent solubility in water, making it especially reliable for use in aqueous solutions. Its stability and sensitivity make it ideal for laboratory applications involving strong acid–base reactions, particularly in titrations of mineral acids. Methyl Orange Disodium Salt Extra Pure is a trusted choice for consistent and accurate results in both educational and professional lab settings.


 Preservatives(food)
Preservatives(food) Flavor Enhancers
Flavor Enhancers Acidulants
Acidulants Sweeteners
Sweeteners Antioxidants
Antioxidants Colorants(food)
Colorants(food) Nutraceutical Ingredients (food)
Nutraceutical Ingredients (food) Nutrient Supplements
Nutrient Supplements Emulsifiers
Emulsifiers
 Collectors
Collectors Dust Suppressants
Dust Suppressants Explosives and Blasting Agents
Explosives and Blasting Agents Flocculants and Coagulants
Flocculants and Coagulants Frothers
Frothers Leaching Agents
Leaching Agents pH Modifiers
pH Modifiers Precious Metal Extraction Agents
Precious Metal Extraction Agents
 Antioxidants(plastic)
Antioxidants(plastic) Colorants (Pigments, Dyes)
Colorants (Pigments, Dyes) Fillers and Reinforcements
Fillers and Reinforcements Flame Retardants
Flame Retardants Monomers
Monomers Plasticizers
Plasticizers Polymerization Initiators
Polymerization Initiators Stabilizers (UV, Heat)
Stabilizers (UV, Heat)
 Antifoaming Agents
Antifoaming Agents Chelating Agents
Chelating Agents Coagulants and Flocculants
Coagulants and Flocculants Corrosion Inhibitors
Corrosion Inhibitors Disinfectants and Biocides
Disinfectants and Biocides Oxidizing Agents
Oxidizing Agents pH Adjusters
pH Adjusters Scale Inhibitors( water)
Scale Inhibitors( water)
 Antioxidants(cosmetic)
Antioxidants(cosmetic) Emollients
Emollients Fragrances and Essential Oils
Fragrances and Essential Oils Humectants
Humectants Preservatives
Preservatives Surfactants(cosmetic)
Surfactants(cosmetic) Thickeners
Thickeners UV Filters
UV Filters
 Fertilizers
Fertilizers Soil Conditioners
Soil Conditioners Plant Growth Regulators
Plant Growth Regulators Animal Feed Additives
Animal Feed Additives Biostimulants
Biostimulants Pesticides (Herbicides, Insecticides, Fungicides)
Pesticides (Herbicides, Insecticides, Fungicides)
 Active Pharmaceutical Ingredients (APIs)
Active Pharmaceutical Ingredients (APIs) Excipients
Excipients Solvents(pharmaceutical)
Solvents(pharmaceutical) Antibiotics
Antibiotics Antiseptics and Disinfectants
Antiseptics and Disinfectants Vaccine Adjuvants
Vaccine Adjuvants Nutraceutical Ingredients (pharmaceutical)
Nutraceutical Ingredients (pharmaceutical) Analgesics & Antipyretics
Analgesics & Antipyretics
 Analytical Reagents
Analytical Reagents Solvents(lab)
Solvents(lab) Chromatography Chemicals
Chromatography Chemicals Spectroscopy Reagents
Spectroscopy Reagents microbiology-and-cell-culture-reagents
microbiology-and-cell-culture-reagents Molecular Biology Reagents
Molecular Biology Reagents Biochemical Reagents
Biochemical Reagents Inorganic and Organic Standards
Inorganic and Organic Standards Laboratory Safety Chemicals
Laboratory Safety Chemicals Specialty Laboratory Chemicals(Special Laboratory Equipment)
Specialty Laboratory Chemicals(Special Laboratory Equipment)
 Demulsifiers
Demulsifiers Hydraulic Fracturing Fluids
Hydraulic Fracturing Fluids Scale Inhibitors(oil)
Scale Inhibitors(oil) Surfactants(oil)
Surfactants(oil) Drilling Fluids
Drilling Fluids
 Dyes and Pigments
Dyes and Pigments Bleaching Agents
Bleaching Agents Softening Agents
Softening Agents Finishing Agents
Finishing Agents Antistatic Agents
Antistatic Agents
 Admixtures
Admixtures Waterproofing Agents
Waterproofing Agents Sealants and Adhesives
Sealants and Adhesives Curing Compounds
Curing Compounds Concrete Repair Chemicals
Concrete Repair Chemicals Anti-Corrosion Coatings
Anti-Corrosion Coatings
 Surfactants(cleaning)
Surfactants(cleaning) Builders
Builders Enzymes
Enzymes Solvents (Cleaning)
Solvents (Cleaning) Fragrances
Fragrances
 Electronic Chemicals
Electronic Chemicals Catalysts
Catalysts Lubricants
Lubricants Photographic Chemicals
Photographic Chemicals Refrigerants
Refrigerants Automotive chemicals
Automotive chemicals Pyrotechnic Chemicals
Pyrotechnic Chemicals
 Biodegradable Surfactants
Biodegradable Surfactants Bio-based Solvents
Bio-based Solvents Renewable Polymers
Renewable Polymers Carbon Capture Chemicals
Carbon Capture Chemicals Wastewater Treatment Chemicals
Wastewater Treatment Chemicals
 Pigments
Pigments Solvents(paint)
Solvents(paint) Specialty Coatings
Specialty Coatings Binders/Resins
Binders/Resins Additives
Additives Driers
Driers Anti-Corrosion Agents
Anti-Corrosion Agents Functional Coatings
Functional Coatings Application-Specific Coatings
Application-Specific Coatings
 Leavening Agents
Leavening Agents Dough Conditioners
Dough Conditioners Flour Treatments
Flour Treatments Fat Replacers
Fat Replacers Decoratives
Decoratives Preservatives(baking)
Preservatives(baking)
 Plasticizers & Softeners
Plasticizers & Softeners Reinforcing Agents
Reinforcing Agents Adhesion Promoters
Adhesion Promoters Vulcanizing Agents
Vulcanizing Agents Antidegradants
Antidegradants Blowing Agents
Blowing Agents Fillers & Extenders
Fillers & Extenders Accelerators & Retarders
Accelerators & Retarders